Add:No. 369 Hedong Rd. Hi-tech Industrial Development Zone, 266112, Qingdao, Shandong, China
E-mail:sales@hightopbio.com

to Use

Reaction

Accuracy
The SARS-CoV-2/Flu A+B/RSV/ADV/MP Antigen Rapid Test is intended for invitro qualitative detection to severe acute respiratory syndrome coronavirus2 (SARS-CoV-2) antigen, influenza A+B (Flu A+B) antigen, respiratory syncytialvirus (RSV)antigen, respiratory adenovirus (ADV)and Mycoplasmapneumoniae (MP) antigen in samples from the human anterior nasal cavity area.
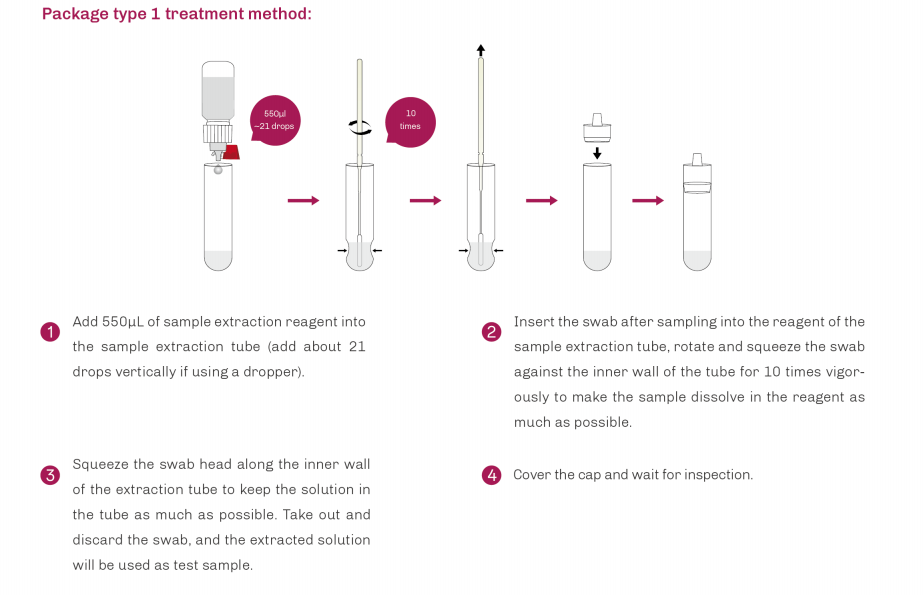
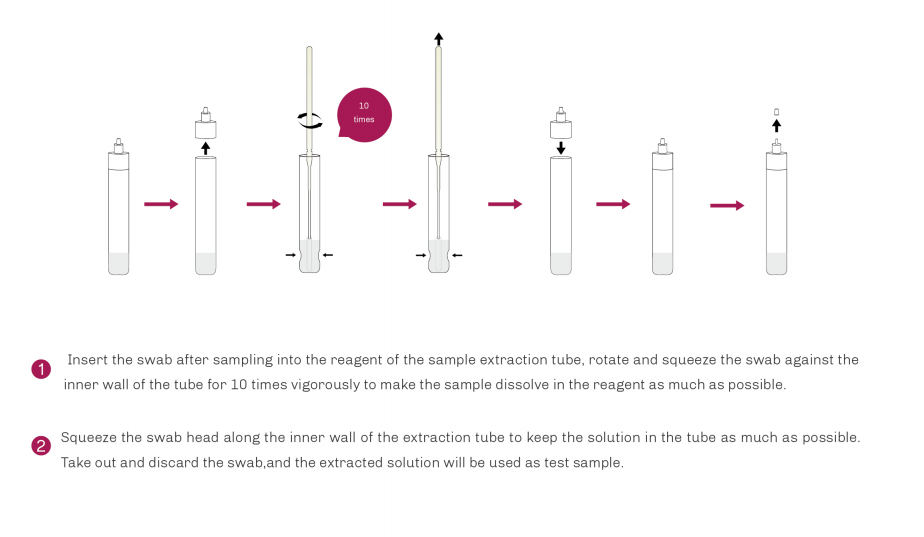
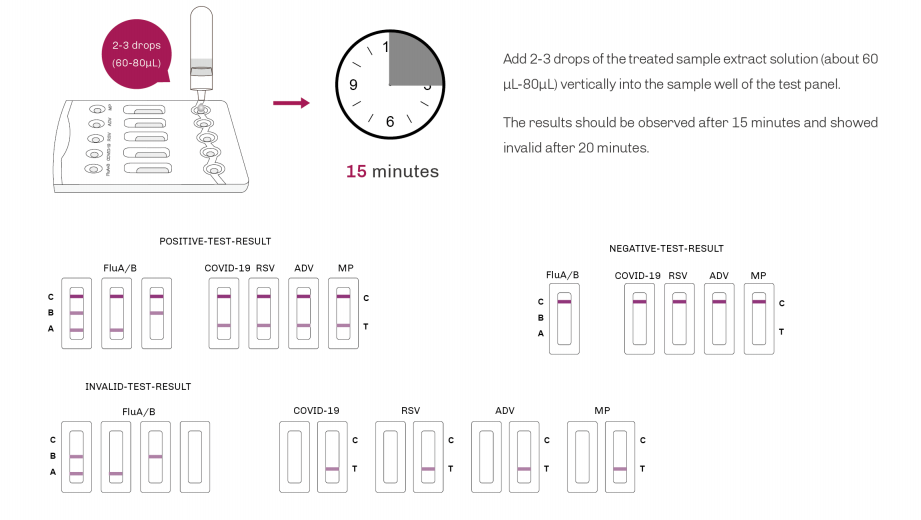
| Product Description | Format | Sample Type | Packing | Validity | LoD | Certification |
SARS-CoV-2/Influenza A+B/RSV/ADV/MP Antigen Rapid Test | Cassette | Nasopharyngeal swab Oropharyngeal swab | 20T | 24 Months | SARS-CoV-2: 8 TCID50/L Flu A: 3.2×104TCID50/L Flu B: 5.2×105TCID50/L RSV: 1.0×102TCID50/L ADV: Type3 6×103 VP/mL Type 7 1×104 VP/mL MP: 6.2×103CFU/mL | CE |
